Policy Benefits Unleashed: Classification Adjustments for 31 Classes of Medical Devices—Dewei Medical Supports Industry Innovation and Upgrading
Release date:
2026-01-29 11:01
On January 4, 2026, the National Medical Products Administration officially issued an announcement on the reclassification of medical devices, implementing optimized classification adjustments for 31 categories of medical devices across 10 subdirectories, covering such high-potential areas as minimally invasive intervention, rehabilitation aids, and medical consumables. This reclassification is firmly grounded in the core principles of “risk-based alignment, science-based regulation, and innovation promotion,” which not only further refine the medical device regulatory system and effectively ensure the safety and efficacy of medical devices used by the public, but also specifically ease regulatory burdens and create a more conducive environment for innovation and development in the medical device industry—particularly by removing barriers to growth for innovative companies focused on the field of minimally invasive intervention. The move delivers clear policy support benefits, propelling the industry’s high-quality development into a new phase.

Image source: National Medical Products Administration website
The core highlights of this revision to the medical device classification system are concentrated in three key areas, each of which is precisely aligned with industry development needs and addresses the real pain points faced by enterprises. First, low-risk products are downgraded to reduce regulatory burdens: specifically, previously Class II low-risk devices such as delivery guidewires and manual wheelchairs are now reclassified as either Class I or Class II for filing-based management. This significantly streamlines the registration and approval process, shortens the registration timeline, lowers compliance costs and R&D investment, and helps these commonly used clinical products enter the market more quickly to serve a broad patient population. Second, high-risk products are subject to more precise control: for high-risk devices in fields such as minimally invasive intervention and in vitro diagnostics, the classification criteria have been further refined, product categorization has been clarified, and end-to-end regulatory requirements have been strengthened, effectively closing regulatory loopholes and ensuring the safe clinical use of high-risk medical devices. Third, the classification standards are being aligned with international practices: the revisions draw extensively on the classification guidelines of the International Medical Device Regulators Forum (IMDRF), standardizing classification criteria and reducing technical barriers and approval hurdles for multinational registration of domestically produced medical devices, thereby helping high-quality Chinese-made devices expand into global markets and compete on the world stage. Notably, for companies specializing in minimally invasive interventional devices, this classification adjustment not only accelerates the pace of innovation and product iteration but also raises the bar for product testing and validation—precise testing and validation being critical to achieving regulatory compliance and gaining a competitive edge in the market. Leveraging its own technological strengths, Xi’an Dewei Medical is well positioned to provide comprehensive, specialized support services to such companies.
DeWei Medical Products Receive Positive Market Feedback
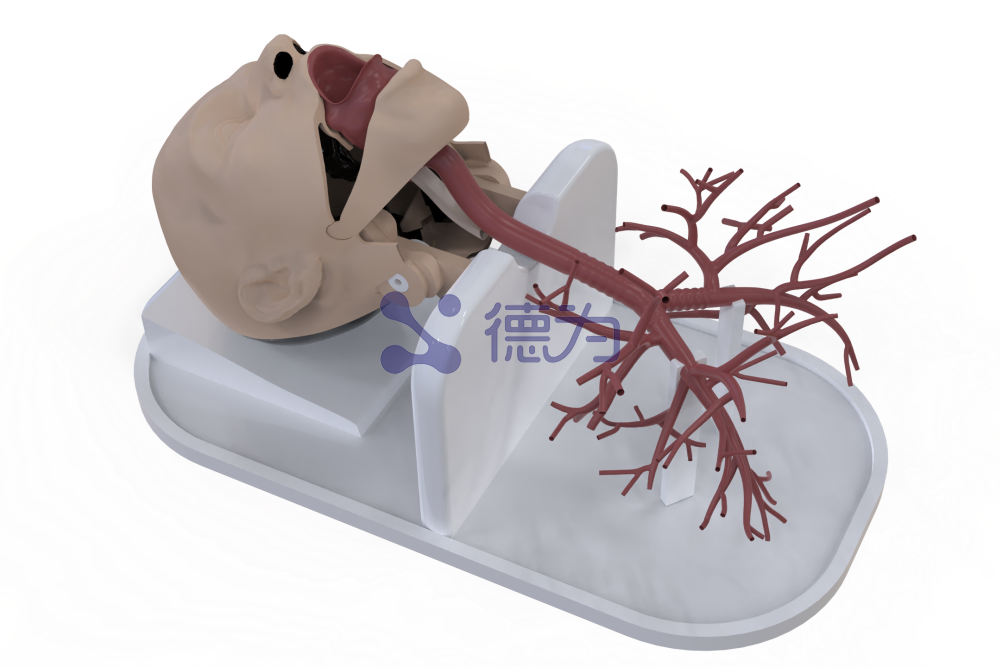
As a leading enterprise in high-end medical testing equipment and solutions, Xi’an Dewei Medical has consistently focused on the core simulation and testing needs of medical device engineers, with years of deep expertise in the development of medical simulation and testing systems, resulting in a robust technological foundation and extensive industry experience. To address the testing needs of minimally invasive interventional medical devices, Dewei Medical has developed a core simulation model that precisely replicates human anatomy on a 1:1 scale. The vascular model is crafted from a proprietary, highly transparent, soft silicone material that not only closely resembles the appearance of human blood vessels but also accurately reproduces their elasticity, resilience, and hemodynamic characteristics, thereby simulating a wide range of complex conditions encountered in real-world clinical procedures. In addition, this simulation testing system is equipped with a comprehensive suite of auxiliary instruments, including high-precision sensors, real-time flow and pressure monitors, and push-force testing devices, enabling precise evaluation of minimally invasive interventional medical devices such as stents, catheters, and guidewires across multiple critical dimensions—including product performance, safety, and biocompatibility. Dewei Medical offers a turnkey testing solution that encompasses everything from test protocol design and equipment calibration to the issuance of test reports, providing comprehensive support to manufacturers. With this professional testing system, enterprises can efficiently conduct performance validation and compliance testing throughout the product development process, ensuring precise alignment with the updated compliance requirements following the recent classification adjustments. This significantly shortens the product development cycle, reduces R&D risks, accelerates the time-to-market for innovative products, and enables companies to swiftly capitalize on policy incentives and gain a competitive edge in the market.
DeWei Medical Product Showcase
This adjustment to the classification of Class III medical devices is not merely a change of category; rather, it represents a pivotal national initiative to steer the medical device industry toward “innovative efficiency, regulatory compliance and controllability, and internationalization.” It both establishes clear regulatory boundaries for industry development and opens up vast opportunities for innovative enterprises. Looking ahead, as the revised classification policy is fully implemented, the medical device sector will usher in a new wave of innovation and upgrading. Competition in hot areas such as minimally invasive intervention will intensify, making rigorous testing and validation, coupled with uncompromising product quality, critical determinants of corporate competitiveness. Xi’an Dewei Medical will continue to leverage its technological strengths in medical simulation and test-system development, relentlessly iterating and enhancing its products and services. The company will provide professional, efficient, and precise testing support to minimally invasive intervention device manufacturers and high-end research laboratories, helping industry players capitalize on policy benefits, overcome technical bottlenecks, and drive high-quality innovation and upgrading across the Chinese medical device sector. In doing so, Dewei Medical will contribute to greater breakthroughs for domestically produced medical devices in both domestic and international markets.
Keywords:
3D printing,Medical devices,Minimally Invasive Intervention,Test equipment,Test System,Medical Simulation,Drug Administration,Neurointervention,Silicone vascular model
Other news
Request a quote
*Please keep your phone accessible—we'll reach out to you within 24 hours.