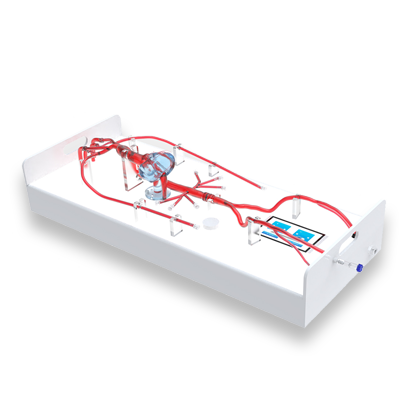
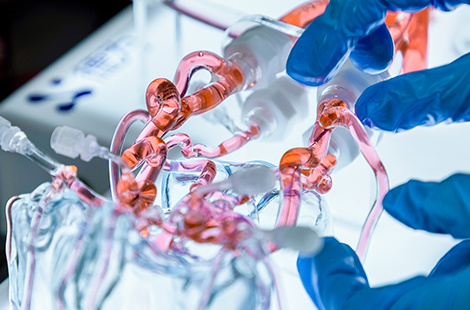
Simulation Capabilities

One-click Shadowless Illumination
Built-in illuminated panel ensures even lighting, making instruments clearly visible.

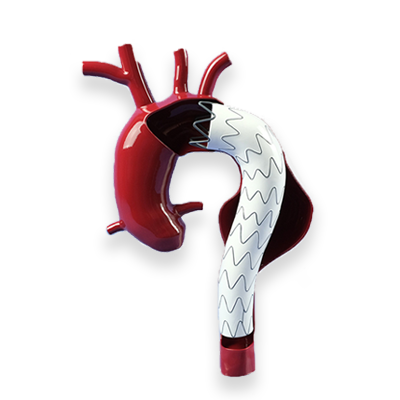
Customizable Lesion Modules
Customizable structural, functional, inflammatory, thrombotic, and tumoral lesions
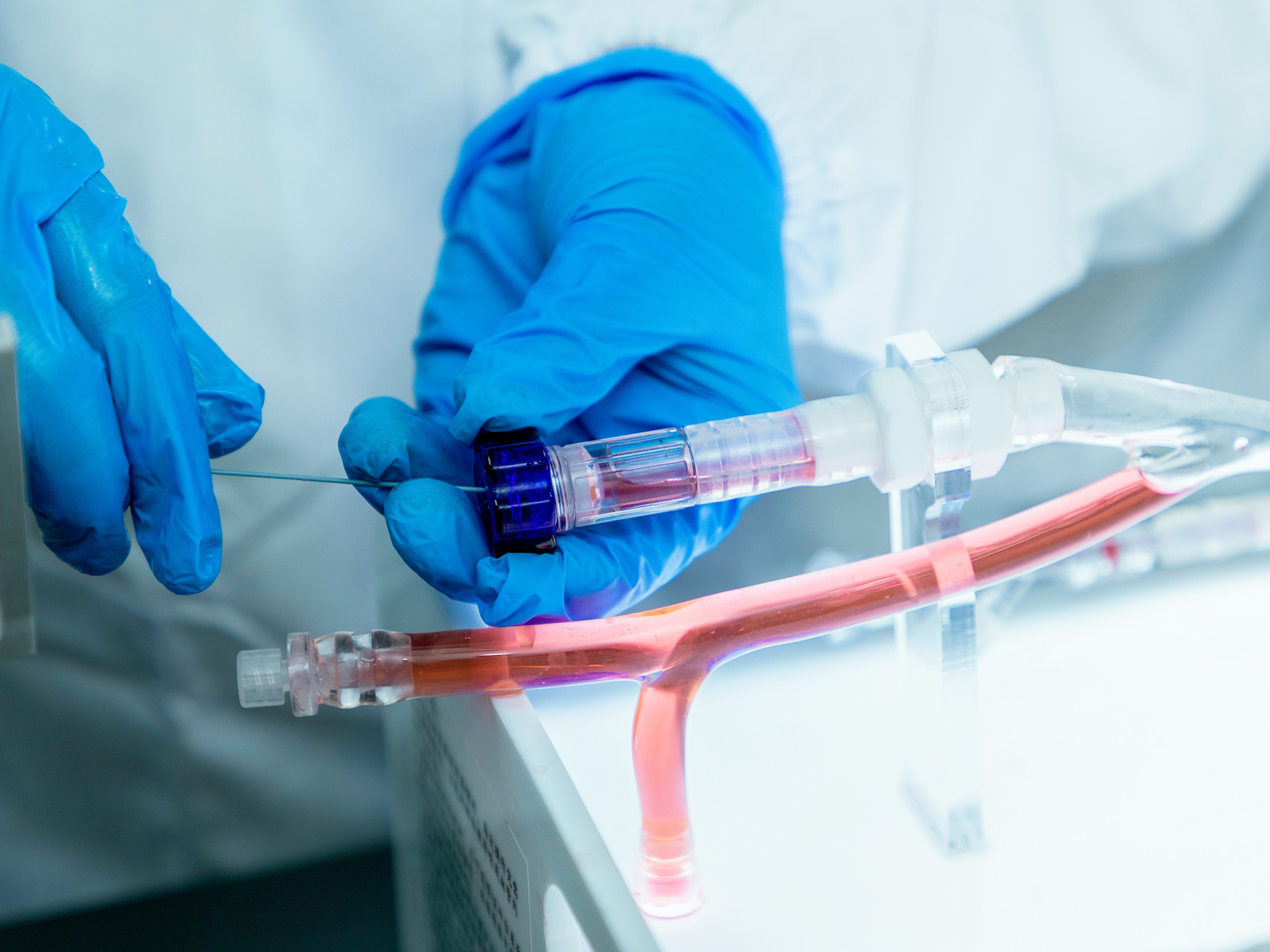
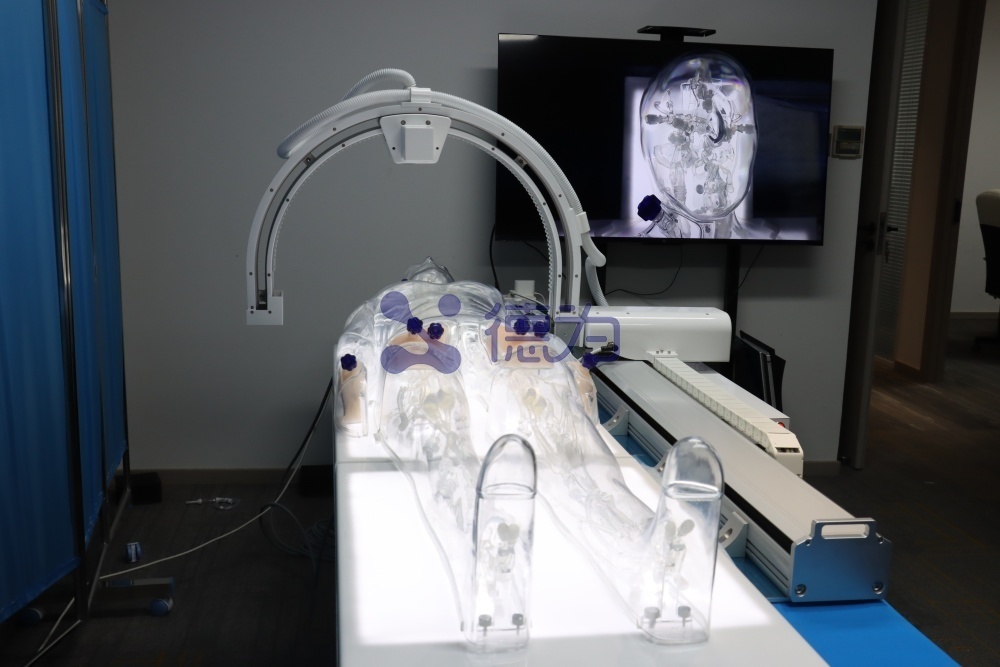
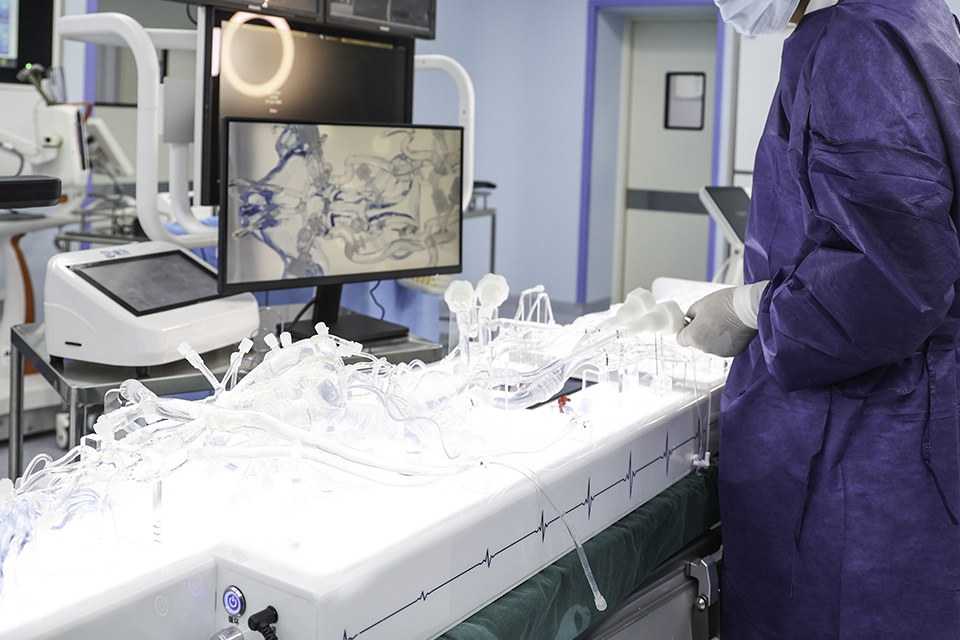
Multi-procedure Interventional Simulation
Balloon angioplasty, stenting, embolization, thrombolysis/thrombectomy, and filter placement
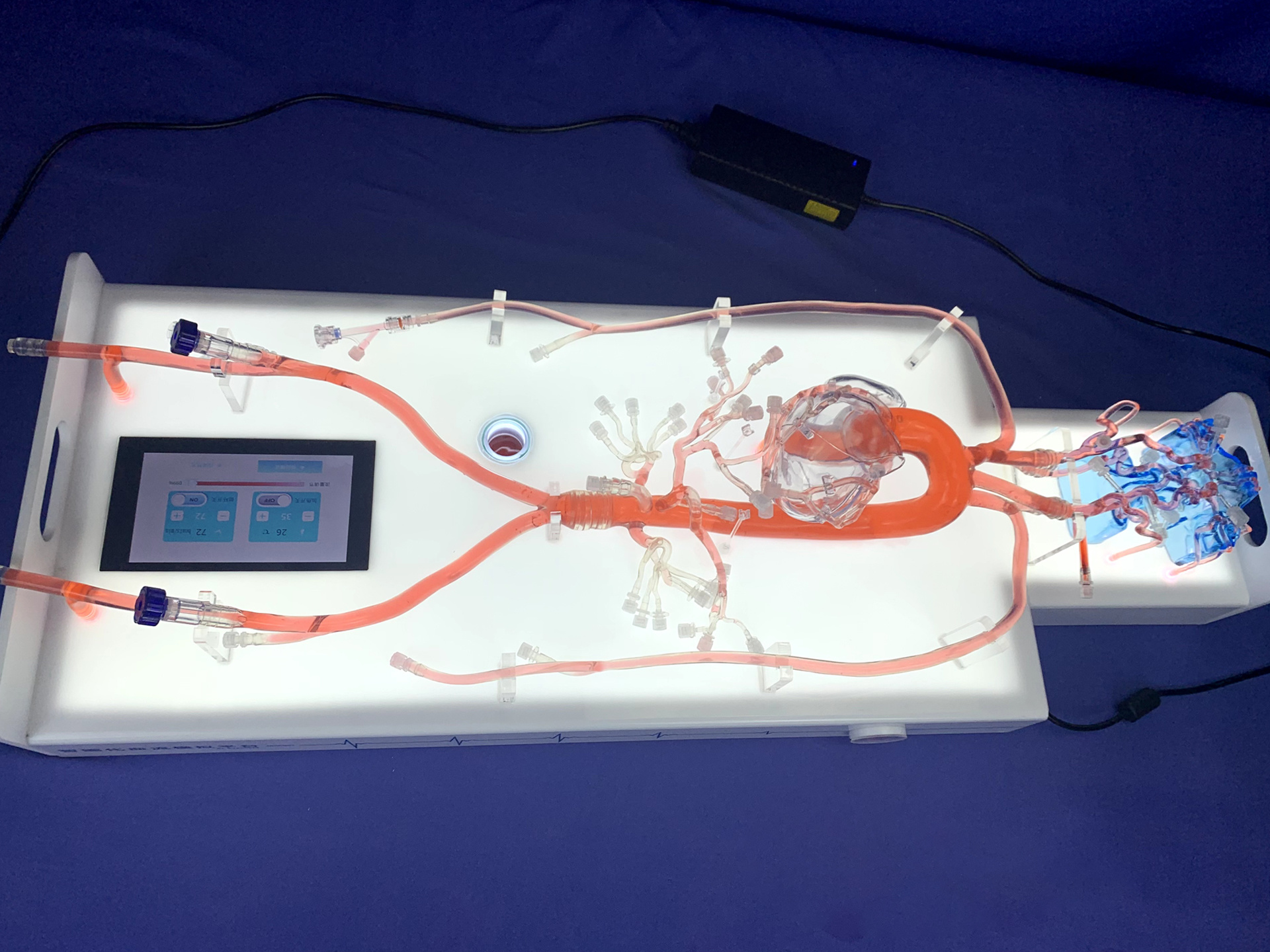
Integrated All-in-One Design
Integrates pipelines, pump body, control components, and the lighting system into a single unit—no messy piping involved.

Intelligent Flow Control
External HD touch screen allows adjustable blood flow parameters such as flow rate and pulsation.
PRODUCT CENTER
Products


SHOWCASE
Showcase
2025
12-25
2025
12-12
2025
11-21
2025
11-13
ABOUT US
About Us