Push Force Testing Device/Guidewire and Catheter Push Force Testing Device
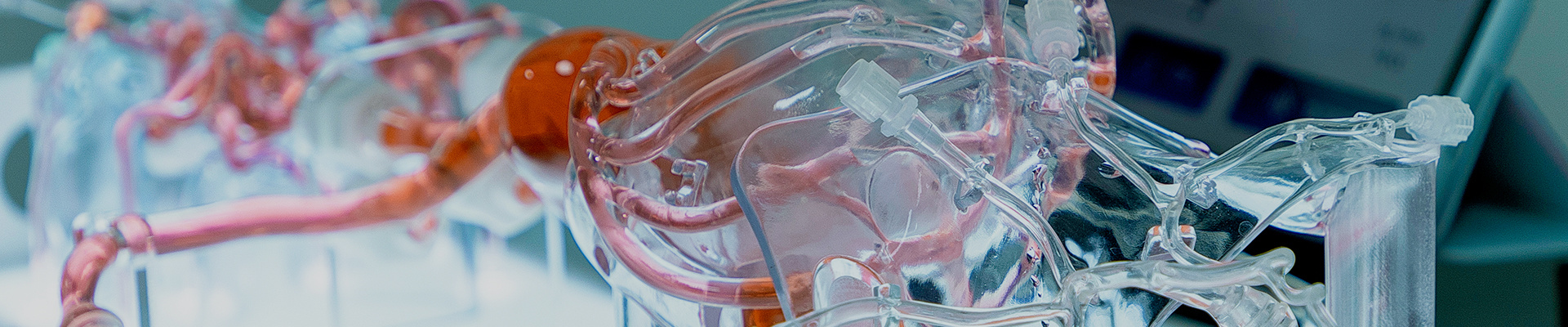
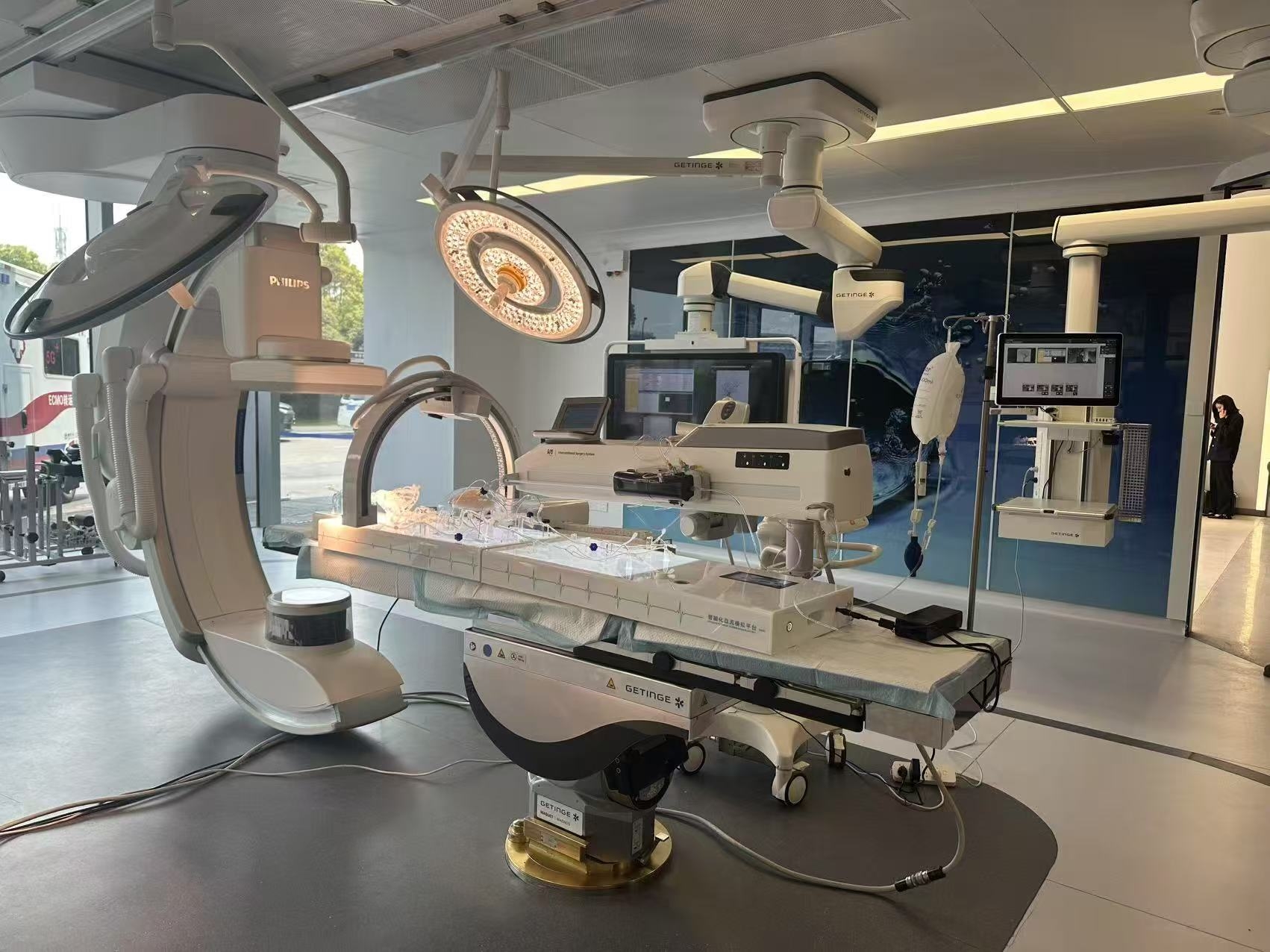
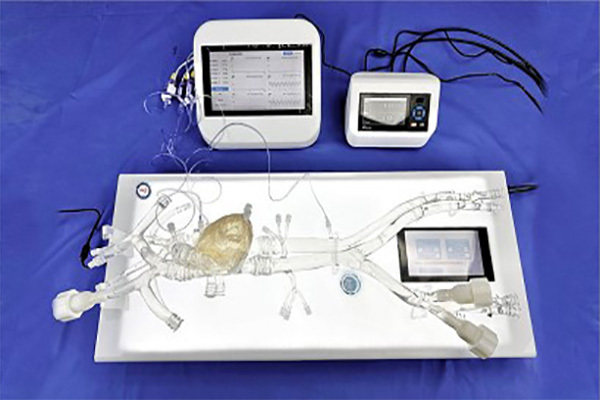
The Guidewire and Catheter Delivery and Rotation Testing System is designed and developed for the evaluation and research of interventional devices used in cardiovascular disease treatment. The system consists of a push force detection device and a medical simulation model, providing a highly realistic experimental environment for performance testing of interventional instruments and surgical training. In addition, the system supports customized services, allowing testing with different vascular model structures according to client needs to meet diverse testing requirements. The testing system complies with international standards such as YY/T 0872-2013, ensuring the accuracy and standardization of test results.
Market price:
Retail price:
Inventory remaining:
Specialty:
Push Force Testing Device/Guidewire and Catheter Push Force Testing Device
Keywords:
testing
interventional
guidewire
Email: sales@deweimed.com
隐藏域元素占位
Push Force Testing Device/Guidewire and Catheter Push Force Testing Device
The Guidewire and Catheter Delivery and Rotation Testing System is designed and developed for the evaluation and research of interventional devices used in cardiovascular disease treatment. The system consists of a push force detection device and a medical simulation model, providing a highly realistic experimental environment for performance testing of interventional instruments and surgical training. In addition, the system supports customized services, allowing testing with different vascular model structures according to client needs to meet diverse testing requirements. The testing system complies with international standards such as YY/T 0872-2013, ensuring the accuracy and standardization of test results.
Category:
Push Force Testing Device/Guidewire and Catheter Push Force Testing Device
Keywords:
testing
interventional
guidewire
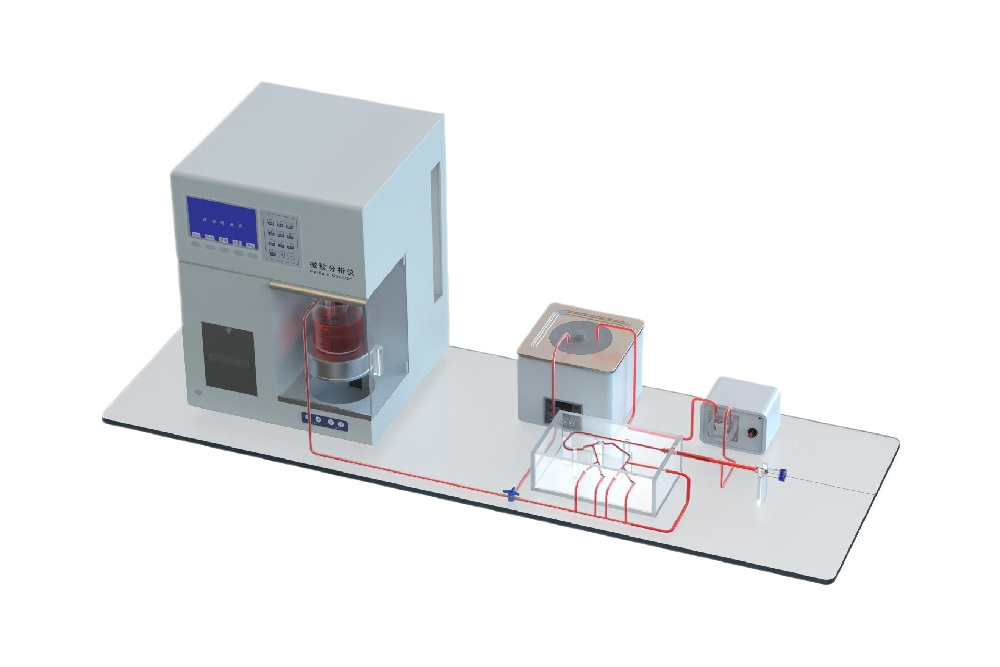
Technical Highlights




Product Features

Built-in blood flow simulation system
Simulates vascular environments under different temperatures and hemodynamic conditions, enhancing testing accuracy and reliability.

Visualized operation
Integrated large-area, high-brightness backlight unit with high-definition camera ensures clear visibility of interventional device testing, allowing easy observation and timely adjustments.

Precise force control
Equipped with high-sensitivity force and torque sensors with ultra-small ranges, capable of detecting minimum force changes as low as 0.001 N. The motor with reduction system ensures stability and efficiency during testing.

Control system
Computer software provides real-time curve analysis, facilitates data processing and transmission, and automatically generates test reports.
Structure
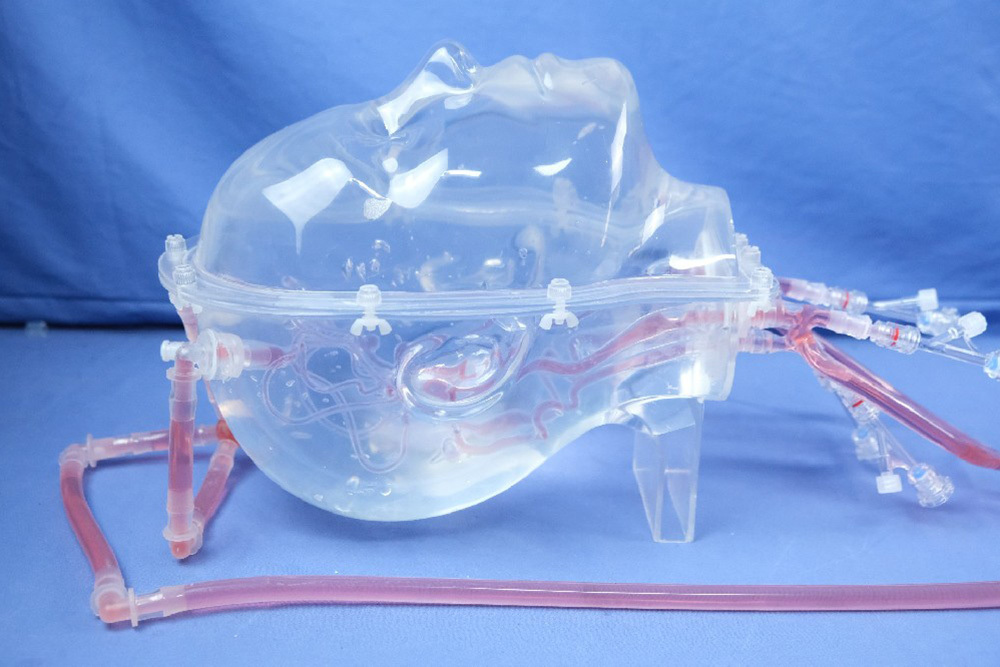
Interventional Device Push Force Testing Equipment: The Guidewire and Catheter Push-and-Rotation Testing System is a specialized device designed to evaluate the performance of interventional medical instruments (such as guidewires, catheters, and sheaths) in terms of pushability and torquability within vascular environments. It primarily simulates the manipulation of devices during clinical procedures, testing their mechanical characteristics, flexibility, and reliability.
Dimension Drawing

Customization
Model Customization: Customizable cardiovascular and arteriovenous models
Accessories Customization: High-definition cameras, force sensors, reduction motors, and clamping systems can be customized according to customer requirements
Software Customization: Data analysis and processing can be tailored to customer testing needs, with report generation included
Accessories

Proximal Push-Back Force Measurement Module

Proximal Torque Measurement Module

Remote Torque Measurement Module
Parameters
Type | Push Force Testing Device/Guidewire and Catheter Push Force Testing Device |
Image |  |
Model | PTFT001 |
Proximal Push/Pull Force Range | -30N-30N |
Sensor CCC accuracy | ±0.02% |
Minimum force accuracy | ±0.001N |
Proximal Torque Sensor Range | -0.5N~0.5N |
Proximal Torque Sensor Accuracy | ±0.2%F.S |
Proximal Minimum Torque Accuracy | ±0.001Nm |
Sensor Overload Resistance | 120% |
Roller Push/Pull Speed Range | 0~30mm/s |
Proximal Torsional Velocity Range | 0~360°/s |
Distal Torque Sensor Range | -0.5~0.5Nm |
Distal Torque Sensor Accuracy | ±2%F.S |
Distal Minimum Torque Accuracy | ±0.001Nm |
Suitable Products | Various guide wires, catheters, delivery systems, sheaths, etc |
Force Sensor Sampling Rate | 10 o'clock/second |
Specifications | AC220V,50Hz |
External Dimensions | 5.9*2.2*3.6ft |
Weight | 220.5lb |
Test Report
Related News
Related videos

Full-Body Arteriovenous Intervention Vascular Model – Flagship Edition

Biomimetic DSA System (C-arm)

Neurointerventional Surgery Simulation System

Lower Limb Guidewire Passability Test Model
Request a quote
*Please keep your phone accessible—we'll reach out to you within 24 hours.